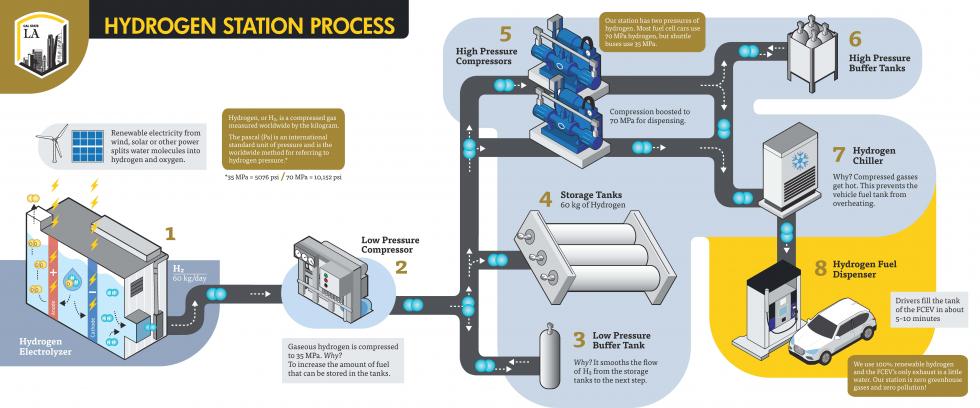
The hydrogen at Cal State LA is produced using a process called electrolysis, where the water molecule is split into oxygen and hydrogen using electricity. The processed hydrogen is then compressed up to 6,200 psi and placed in storage tanks. During vehicle fueling, the hydrogen is compressed again, this time up to 10,000 psi.
Hydrogen has a good energy density by weight but poor energy density by volume, so the compression increases the density in the same volume, allowing more energy to be delivered. The pressure of 5,000 psi allows the consumer to fuel to slightly more than half a tank, while the 10,000 psi will result in a full tank.
The dispenser has a screen indicating how many kilograms of hydrogen was delivered to the car and the price to be paid, just like gasoline dispensers.
The dispenser nozzle is equipped with infrared technology that communicates information to a central controller, like the vehicle tank temperatures and pressures. This information allows the system to plan the fueling rate, to know when fueling is completed, and if any problems occur, the process can be stopped automatically.
At first, fueling starts from the storage tanks without the aid of compressors. As the pressure between the storage tanks and the vehicle tanks reaches equilibrium at approximately 6000 psi, the high-pressure compressors start operation and top off the vehicle tanks to over 10,000 psi. In practice, the target pressure might even exceed 11,000 psi as the hydrogen entering the tank heats up.
When hydrogen in the vehicle tanks eventually cools off to ambient temperature, the pressure also drops, providing about 95-98% of the tank's design capacity. This is also known as the tank's "state of charge" (SOC).
During fueling, the hydrogen is also passed through a chiller unit operating at about -36°C with the goal being that most of the hydrogen entering the vehicle is at about -17°C to -20°C (Type B fueling). The precooling of the hydrogen allows for faster fueling times. Some stations might have their chiller units set below -40°C (Type A fueling), to perform fueling even faster.
The process of fueling with hydrogen was designed to be similar to CNG fueling, making it easier for the consumer. The picture below illustrates the fuelling components.
Yes, fueling with hydrogen is completely safe. This technology has been used in different fields for many years and is now applied to vehicles. The station is equipped with leak sensors, flame detectors, and other safety devices that constantly monitor the fueling process. One such measure is a leak test performed every 3,000 psi pressure increase during fueling to check for leaks in the fueling hose, nozzle, and vehicle receptacle. The system periodically pauses and closes the hydrogen inlet valve to the dispenser to perform this test. If no measurable drop in pressure is detected during a 5-second pause, this indicates the absence of leaks, and fueling is allowed to resume to completion.
Please contact us with questions or inquiries about the Cal State LA Hydrogen Fueling and Research Facility.
Dr. David Blekhman at [email protected] or Blake Cortis at [email protected]