- Protein–Ice Interactions for Enzyme and Cell Protection
- New Roles of Antifreeze Proteins - Antifreeze Proteins as Highly Effective Additives to Control the Crystallization of Non-Ice like Compounds
- Antifreeze Protein Enhancers: Identification and Mechanisms
- Effects of Toxic Metal Ions on Enzymes and Mammalian Cells
- Collaborators
- Funding
Protein–Ice Interactions for Enzyme and Cell Protection
Antifreeze proteins (AFPs), a class of ice binding proteins (IBPs), mitigate freeze induced damage by binding to ice, suppressing recrystallization, and modulating nucleation and growth. Our work shows that AFPs provide dual protection for enzymes during freezing: they stabilize enzymatic structure and activity while inhibiting ice recrystallization to reduce stress on biomolecules (Int. J. Biol. Macromol. 2026, 2019).
At the cellular scale, beetle AFPs reduce cold‑induced swelling and improve survival, while toxicological studies in mammalian models establish both safety and protective effects, supporting translational potential in biomedical and biopharmaceutical applications (Biomolecules 2022; Toxicology Lett. 2022).
High‑resolution synchrotron X‑ray diffraction further differentiates protein–ice interfacial behaviors among pharmaceutical proteins, sharpening structure–function principles for rational design (JPC 2019).
Building on these insights, we aim to develop robust bio‑antifreeze systems for long‑term preservation of enzymes, biologics, cells, and tissues. We integrate molecular engineering with quantitative assays of ice recrystallization, enzyme activity, and mammalian cell stress responses, linking molecular recognition to cell preservation outcomes across molecular, cellular, and system scales.
(top)
New Roles of Antifreeze Proteins - Antifreeze Proteins as Highly Effective Additives to Control the Crystallization of Non-Ice like Compounds
Antifreeze proteins and glycoproteins [AF(G)Ps] have been well-known for their ability to inhibit ice growth and recrystallization by interactions with specific ice crystal faces, representing a noteworthy example of crystal control in nature. Recently, we have demonstrated that AF(G)Ps can also control the growth of non-ice like crystalline solids, such as nucleosides and sugars, in a highly effective manner, that is the molar ratio can be used as low as 10-7. This newly identified role of AF(G)Ps further expands the molecular recognition repertoire of AF(G)Ps. Crystallization control is crucial in various fields, e.g., in pharmaceutical and food industries. Our goals are to understand on the molecular recognition of non-ice like crystalline solids by AF(G)Ps in details and to finally have extensive applications of AF(G)Ps into industries.
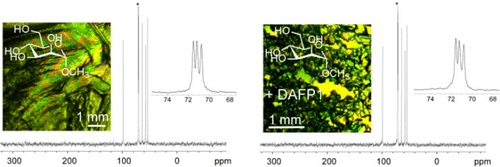
The crystal morphology of methyl α-D-mannopyranoside (MDM) crystals can be effectively controlled by antifreeze proteins (JACS, 2014 (link), 136, 8973-8981; C&EN, 2014, 92, 22 (link)).
The crystallization of 5-methyluridine can be effectively controlled by antifreeze proteins (Chem. Commun. 2012,48, 11555-11557 (link)).
We also demonstrate that molecular control of crystallization is central to effective biopreservation. AFPs as biopolymer additives can completely inhibit polyol nucleation. They regulate trehalose precipitation in freezing‑avoiding insects, revealing how recognition governs excipient phase behavior at low temperature (Cell Rep. Phys. Sci. 2022; PNAS 2016).
(top)
Antifreeze Protein Enhancers: Identification and Mechanisms
As an adaptive response to cold environmental conditions, many organisms, such as fish, plants and insects, have developed antifreeze proteins (AFPs) that allow them to survive at subfreezing temperatures. AFPs exhibit great structural diversity, while they are all characterized by their unique abilities to lower the freezing point of water without affecting the melting point. The difference between the nonequilibrium freezing and melting points, termed thermal hysteresis (TH), is usually used as a measure of the activity of AFPs. The details of their mechanism remain elusive. AFPs have many potential applications in biomedical and other fields, such as in cryopreservation and food industry. Highly efficient enhancers can greatly increase the antifreeze activity of AFPs and thus reduce the cost of the usage of AFPs in these applications. Many low molecular mass molecules and other co-solutes have been identified as enhancers for some AFPs, however, the molecular mechanism of enhancement is still lacking.
We aim to understand the molecular mechanisms of the AFP enhancers and the enhancing function of AFPs. To approach the aims, we correlate the enhancement abilities of the enhancers with their varying physicochemical properties as well as we investigate the interactions between AFP enhancers and AFPs by integrating the molecular biological techniques and various biophysical and biochemical methods.
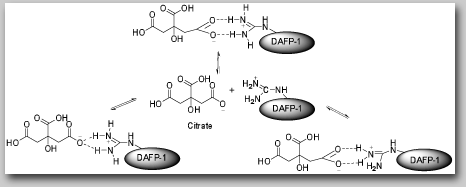
Proposed interactions between an isoform of AFP from the beetle Dendroides canadensis
(DAFP-1) and citrate, a strong AFP enhancer (Biochemistry, 2009, 48, 9696-9703). (link-pdf)
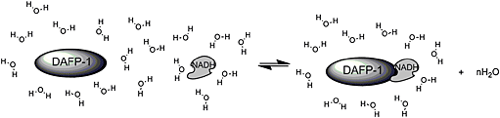
Reduced nicotinamide adenine dinucleotide (NADH) has been identified as a most efficient
enhancer of DAFP-1 (J. Mol. Recognit. 2011, 24, 1025-1032 (link-PDF)
(top)
Effects of Toxic Metal Ions on Enzymes and Mammalian cells
Zinc fingers (ZnFs) are small protein motifs with one or more Zn(II) as a structural factor and conserved cysteine and/or histidine residues as ligands. The Zn(II), tetrahedrally coordinated by the side chains of the ligands, is crucial for the function of the ZnF-containing protein. ZnF-containing proteins are among the most abundant protein superfamilies in eukaryotic genomes and participate in a variety of cellular processes, such as replication and repair, transcription and translation, and metabolism and signaling, where they typically mediate sequence-specific nucleic acid binding, protein-protein interactions, and protein-lipid interactions. ZnFs have been proposed to be cellular targets for the carcinogenic metals having high affinities for sulfhydryl groups that are increasingly available in the environment due to the industrial discharge of waste, e.g., Pb(II), Hg(II), and Cd(II). The interactions of the carcinogenic metal ions with ZnFs have been proposed to distort or damage the structure of ZnFs and thus may induce a number of adverse physiological effects on human health including cancers, reproductive and developmental defects, and neuropathies.
Our goal is to determine the detailed structure basis of the interactions of toxic metal ions with ZnF-containing DNA repair proteins. The atomic-level investigation of the toxic metal-bound sites are crucial to understand the relationship between the altered structure upon metal binding and metal carcinogenesis. A better understanding of molecular mechanisms of metal carcinogenesis will be obtained.
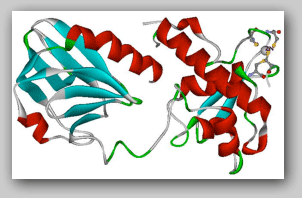
Tertiary structure of the bacterial formamidopyrimidine-DNA glycosylase (Fpg)
The ZnF is locatedat C-terminus of the protein. The zinc is shown as a ball in grey
and the four Cys ligands shown as a ball and stick model.
(top)
Collaborators
Prof. John G. Duman (University of Notre Dame)
Prof. William A. Goddard III (California Institute of Technology)
Prof. Timothy O'Connor (City of Hope)
Dr. Sen Wang (Stanford University)
Prof. Arnold L. Rheingold (University California, San Diego)
Prof. James A. Golen (University of Massachusetts, Dartmouth)
Prof. Arthur L. DeVries (University of Illinois at Urbana–Champaign)
Prof. Ravinder Abrol (CSUN)
Dr. Evgenyi Shalaev (Abbvie)
Prof. Keiko Omori (City of Hope)
Prof. Martine Culty (University of Southern California)
Prof. Tod A. Pascal (University of California, San Diego)
Prof. Caitlin Scott (Cal State LA)
Funding
NIH
Research Corporation
CSUBIOTECH (formerly CSUPERB)
ACS-PRF
NSF
RSCA