Microbiology and Immunology
Faculty research interests in the area of microbiology and immunology include fungal pathogenesis (Park Lab), innate mucosal immunity (Porter Lab), microbial ecology (Ramirez Lab), environmental microbiology (Salmassi Lab), applied microbiology and biotechnology (Xu Lab), and Molecular Microbiology (Cohen Lab). An asterisk following a name in a publication citation indicates a student coauthor.
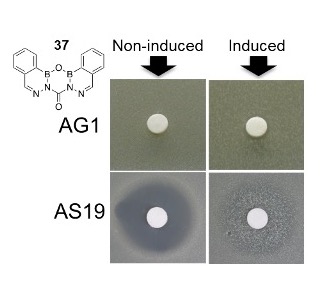
Fungal Pathogenesis
 |
Contact: Hyunsook Park, Ph.D. Office: ASCL 353, ext: 3-2060 Laboratory: ASCL 348, ext: 3-2035 E-mail: [email protected] Web: /faculty/hyunsook-park |
|
| Confocal microscopy of the yeast C. albicans grown with oral epithelial cell. Germ tube (on the right) penetrates into the mammalian cell. |
Research Summary
Our research program focuses on host-pathogen interactions in infectious disease. In particular, we have focused on the pathogenic mechanisms of the fungus Candida albicans during the initiation of oropharyngeal candidiasis. We are identifying key virulence factors governing the early stage of oral candidiasis using a transcriptional profiling analysis and gene disruption approach.
| Representative Publications |
| Park, H., Liu, Y., Solis, N., Spotkov, J., Hamaker, J., Blankenship, J.R., Yeaman, M.R., Mitchell, A.P., Liu, H., and Filler, S.G. 2009. Transcriptional responses of Candida albicans to epithelial and endothelial cells. Eukaryotic Cell (in press). |
| Gank, K.D., Yeaman, M.R., Kojima, S., Yount, N.Y., Park, H., Edwards, Jr., J.E., Filler, S.G., and Fu, Y. 2008. SSD1 is integral to host defense peptide resistance in Candida albicans. Eukaryotic Cell, May 30. |
| Barker, K.S., Park, H., Phan, Q.-T., Xu, L., Homayouni, R., Liu, T.T., Rogers, P.D., and Filler, S.G. 2008. Transcriptome profile of the vascular endothelial cell response to Candida albicans. Journal of Infectious Diseases 198: 193-202. |
| Martinez-Lopez, R., Park, H., Myers, C.L., Gil, C., and Filler, S.G. 2006. Candida albicans Ecm33p is important for normal cell wall architecture and interactions with endothelial and oral epithelial cells. Eukaryotic Cell 5: 140-147. |
| Park, H., Myers, C.L., Sheppard, D.C., Phan, Q.T., Sanchez, A.A., Edwards, Jr., J.E. and Filler, S.G. 2005. Role of the fungal ras-protein kinase A pathway in governing epithelial cell interactions during oropharyngeal candidiasis. Cellular Microbiology 7: 499-510. |
Innate Mucosal Immunity
 |
Contact: Edith Porter, M.D. Office: ASCL 355, ext: 3-6353 Laboratory: ASCL 333, ext: 3-2079 E-mail: [email protected] Web: http://instructional1.calstatela.edu/eporter/porterlab |
|
| Transmission electron microscopy, 10,000-fold magnification, of Pseudomonas aeruginosa showing ultrastructural changes of the cell wall after treatment with the free fatty acid docosahexaenoic acid and the antimicrobial polypeptide lysozyme. |
Research Summary
Our research focuses on mucosal innate immunity, specifically, antimicrobial effector molecules that directly act on and kill bacterial pathogens. A great deal of our work has targeted the activity and regulation of the antimicrobial peptide human defensin 5 in the intestinal and genital tract and the response of Paneth cells, specialized defense cells in the small intestine, to infection with Salmonella. Recently, we have begun to explore role of host-derived antimicrobial lipids, in particular cholesteryl esters, in the mucosal defense of the respiratory and genital tract.
| Representative Publications |
| Rodriguez, N.R.*, Eloi, M.D.*, Huynh, A.*, Dominguez, T.*, Cheung Lam, A.H.*, Carcamo-Molina, D.*, Naser, Z.*, Desharnais, R., Salzman, N.H., and Porter, E. 2012. Expansion of Paneth Cell Population in Response to Enteric Salmonella Infection. Infection and Immunity 80: 266-275. |
| Spencer, J.D., Hains, D.S., Porter, E., Bevins C.L., Dirosario J., Becknell B., Wang H., and Schwaderer A.L. 2012. Human alpha defensin 5 expression in the human kidney and urinary tract. PLoS One 7: e31712. |
| Lee, J.T., Jansen, M.*, Yilma, A.N.*, Nguyen, A.*, Desharnais, R., and Porter, E. 2010. Antimicrobial lipids: novel innate defense molecules are elevated in sinus secretions of patients with chronic rhinosinusitis. American Journal of Rhinology & Allergy 24: 99-104. |
| Martinez, J.G.*, Waldon, M.*, Huang, Q.*, Alvarez, S.*, Oren, A., Sandoval, N.*, Du, M., Zhou, F., Zenz, A., Lohner, K., Desharnais, R., and Porter, E. 2009. Membrane-targeted synergistic activity of docosahexaenoic acid and lysozyme against P. aeruginosa. Biochemical Journal 419: 193-200. |
| Do, T.Q., Moshkani, S.*, Castillo, P.*, Anunta, S.*, Pogosyan, A.*, Cheung, A.*, Marbois, B., Faull, K.F., Ernst, W., Chiang, S.M., Fujii, G., Clarke, C.F., Foster, K., and Porter, E. 2008. Lipids including cholesteryl linoleate and cholesteryl arachidonate contribute to the inherent antibacterial activity of human nasal fluid. Journal of Immunology 181: 4177-4187. |
Microbial Ecology
| Contact: Gustavo Ramirez, Ph.D. Office: ASCL 218, ext: 3-7480 Laboratory: ASCR 347 , ext: 3-XXXX E-mail: [email protected] Web: https://www.ramirezlaboratory.com |
||
| Microbial colonization experiment deployed at a deep-sea octopus nursery located ~3km depth in the Costa Rican Pacific, El Dorado Outcrop, summer of 2023. |
Research Summary
We are engaged in interdisciplinary investigations that integrate ecology and genomics to study the diversity, distribution, and functioning of microbial communities in nature. This involves high-throughput molecular sequencing merged with environmental metadata analyzed using high-performance computing. Our work is at the nexus of basic science and applications in conservation biology, environmental monitoring, and biotechnology. Microbes and their activities are agnostic to environments; thus, on-going projects in our group include exploring the ecology of marine, animal GI, and desert soil related microbiomes.
| Representative Publications |
| Ramírez GA, Bar-Shalom, R, Furlan, A, Romeo, R, Gavagnin, M, Calabrese, G, Garber, AI, Steindler L. 2023. Bacterial aerobic methane cycling by the marine sponge-associated microbiome. Microbiome 11, 49. 2023. https://doi.org/10.1186/s40168-023-01467-4. |
|
Ramírez, GA, Keshri, J, Vahrson, I, Garber, AI, Berrang, ME, Cox, NA, González-Cerón, F, Aggrey, SE, Oakley, BB, et al. 2022. Cecal Microbial Hydrogen Cycling Potential Is Linked to Feed Efficiency Phenotypes in Chickens. Front. Vet. Sci. doi: 10.3389/fvets.2022.904698. |
| Ramírez GA, Paraskevi VM, Edgcomb VA, Teske AP. 2021. Environmental factors shaping bacterial, archaeal and fungal community structure in hydrothermal sediments of Guaymas Basin, Gulf of California. PLOS ONE 16 (9): e0256321. https://doi.org/10.1371/journal.pone.0256321 |
| Garber AI*, Ramírez GA*, McAllister S, Orsi W, D’Hondt S. 2021. Cryptic metabolisms in deep anoxic marine sediment. Envir. Microbiol. doi:10.1111/1758-2229.12983. |
|
Ramírez GA, McKay L.J., Fields, M.W., Ravelo, A.C., Teske, A.P. 2020. The Guaymas Basin Subseafloor Sedimentary Archaeome Reflects Complex Environmental Histories. iScience Journal. Vol 23, Issue 9: https://doi.org/10.1016/j.isci.2020.101459. |
Environmental Microbiology
 |
Contact: Tina Salmassi, Ph.D. Office: ASCL 315, ext: 3-2065 Laboratory: ASCL 327, ext: 3-2083 E-mail: [email protected] Web: http://instructional1.calstatela.edu/tsalmas |
|
| Arsenite oxidizing bacterium (Agrobacterium albertimagni strain AOL15) isolated from the surface of aquatic plants at Hot Creek, California. |
Research Summary
Our laboratory is interested in the interactions between microbes and the environment. We use classical microbiology and molecular biology techniques to study the diversity of bacteria in a variety of environments such as soils, natural waters, plant surfaces, and the hindgut of termites. By observing bacteria both in their natural niches and under controlled laboratory conditions, we hope to better understand the chemistry carried out by these microscopic organisms and their role in the biogeochemical cycling of the elements.
| Representative Publications |
| Pech, H.*, Vazquez, M.*, Van Buren, J.*, Shi, L., Salmassi, T.M., Pasek, M.A., and Foster, K. 2011. Elucidating the redox cycle of reduced phosphorus using ion chromatography. Journal of Chromatographic Science 59: 573-581. |
| Pech, H.*, Henry, A.*, Khachikian, C.S., Salmassi, T.M. Hanrahan, G., and Foster, K.L. 2009. Detection of geothermal phosphite using high-performance liquid chromatography. Environmental Science and Technology 43: 7671-7675. |
| Barco, R.A.*, Patil, D.G.*, Xu, W., Ke, L., Khachikian, C.S., Hanrahan, G., and Salmassi, T.M. 2006. The development of iodide-based methods for batch and on-line determinations of phosphite in aqueous samples. Talanta 69: 1292-1299. |
| Salmassi, T.M., Walker, J.J., Newman, D.K., Leadbetter, J.R., Pace, N.R., and Hering, J.G. 2006. Community and cultivation analysis of arsenite oxidizing biofilms at Hot Creek. Environmental Microbiology 8: 50-59. |
| Hanrahan, G., Salmassi, T.M., Khachikian, C.S., and Foster, K.L. 2005. Reduced inorganic phosphorus in the natural environment: significance, speciation and determination. Talanta 66: 435-444. |
Applied Microbiology and Biotechnology
|
|
Contact: Howard Xu, Ph.D. Office: ASCL 356, ext: 3-2188 Laboratory: ASCL 337, ext: 3-2040 E-mail: [email protected] Web: /research/hxu3 |
|
Research Summary
Our long-term objective is to contribute in the fight against bacterial pathogens through the discovery of novel antibiotic therapeutics, the development of rapid pathogen detection technologies, and molecular characterization of bacterial pathogens. This objective will be achieved by employing several modern, novel and complimentary approaches: bacterial functional genomics, microbial physiology, biochemistry, molecular biology, novel assay development, high-throughput screening, and clinical microbiology. Critical to the success of various research projects are the ongoing collaborations with our colleagues on campus, from UCLA, Los Angeles County USC Medical Center, Indiana University, University of Michigan, National Laboratories, and Canada's National Research Council.
| Representative Publications |
| Meng, J.*, Kanzaki, G.*, Meas, D.*, Lam, C.K.*, Crummer, H.*, Tain, J.*, and Xu, H.H. 2012. A genome-wide inducible phenotypic screen identifies antisense RNA constructs silencing Escherichia coli essential genes. FEMS Microbiology Letters 329: 45-53. |
| Yuan, H., Chai, S.C., Lam, C.K.*, Xu, H.H., and Ye, Q.Z. 2011. Two methionine aminopeptidases from Acinetobacter baumannii are functional enzymes. Bioorganic & Medicinal Chemistry Letters 21: 3395-3398. |
| Golanbar, G.D.*, Lam, C.K.*, Chu, Y.M.*, Cueva, C.*, Tan, S.W.*, Silva. I.*, and Xu. H.H. 2011. Phenotypic and molecular characterization of Acinetobacter clinical isolates obtained from inmates of California correctional facilities. Journal of Clinical Microbiology 49: 2121-2131. |
| Xu, H.H., Trawick, J.D., Haselbeck, R.J., Forsyth, R.A., Yamamoto, R.T., Archer, R., Patterson, J., Allen, M., Froelich, J.M., Taylor, I., Nakaji, D., Maile, R., G. C., K., Pilcher, M., Brown-Driver, V., McCarthy, M., Files, A., Robbins, D., King, P., Sillaots, S., Malone, C., Zamudio, C.S., Roemer, T., Wang, L., Youngman, P.J., and Wall, D. 2010. Staphylococcus aureus TargetArray: Comprehensive differential essential gene expression as a mechanistic tool to profile antibacterials. Antimicrobial Agents and Chemotherapy. 54: 3659-3670. |
| Valentine, S.C.*, Contreras, D.*, Tan, S.*, Real, L.J.*, Chu, S., and Xu, H.H. 2008. Phenotypic and molecular characterization of Acinetobacter baumannii clinical isolates from nosocomical outbreaks in Los Angeles County, California. Journal of Clinical Microbiology 46: 2499-2507. |
Molecular Microbiology
| Contact: Susan Cohen, Ph.D. Office: ASCL 354, ext: 3-2091 Laboratory: ASCL 331 E-mail: [email protected] Web: http://cohenlabcsula.weebly.com/ |
||
|
Fluorescence micrographs of cyanobacterial strain Synechococcus elongatus PCC 7942 (red) expressing a YFP-KaiC fusion, showing that circadian clock protein KaiC is localized diffusely throughout the cell during the day and localized to the cell pole at night. |
Research Summary
Circadian rhythms, driven by an internal biological or circadian clock, are nearly ubiquitous and are found in both prokaryotic and eukaryotic organisms. Cyanobacteria are the simplest organisms and only prokaryotes known to have a rigorously tested and robust circadian clock. We use the genetically tractable Synechococcus elongatus PCC 7942 as a premier model organism for studying the molecular details and regulation of the clock. We use a combination of cell biology, genetics, biochemistry and molecular biology to understand how the Kai oscillator functions in the context of the three-dimensional cell.
| Representative Publications |
|
Susan E. Cohen and Susan S. Golden. Circadian rhythms in cyanobacteria. Microbiology and Molecular Biology Reviews 79(4):373-85 (2015) |
|
Chung Sze Luke, Jangir Selimkhanov, Leo Baumgart, Susan E. Cohen, Susan S. Golden, Natalie A. Cookson and Jeff Hasty. A microfluidic platform for long term monitoring of algae in a dynamic environment. ACS Synthetic Biology 5(1):8-14 (2015) |
|
Yong-Gang Chang, Susan E. Cohen, Connie Phong, William K. Myers, Yong-Ick Kim, Roger Tseng, Jenny Lin, Li Zhang, Joseph S. Boyd, Yvonne Lee, Shannon Kang, R. David Britt, Michael J. Rust, Susan S. Golden and Andy LiWang. A protein fold switch joins the circadian oscillator to clock output in cyanobacteria. Science 349(6245):324-8 (2015)†Undergraduate author |
|
Susan E. Cohen, Marcella L. Erb, Joe Pogliano and Susan S. Golden. Best practices for fluorescent microscopy of the cyanobacterial circadian clock. Methods in Enzymology 551:211-21 (2015) |
|
Susan E. Cohen, Marcella L. Erb, Jangir Selimkhanov, Guogang Dong, Jeff Hasty, Joe Pogliano and Susan S. Golden. Dynamic localization of the cyanobacterial circadian clock proteins. Current Biology 24(16):1836-44 (2014) |
Note: ASCL = Wallis Annenberg Integrated Science Complex-Wing A (La Kretz Hall). When calling from off-campus, the area code and prefix for all telephone extensions is (323) 34X-XXXX.